2020 sexual transmitted disease, STD
According to a large-scale STD research survey conducted by scholars from July 2008 to April 2014, we learned that the total number of newly diagnosed STDs: 2389 people,
Chlamydia urethritis: 736 people (30.8%);
Genital herpes: 658 people (27.5%) Type 1: 413 people (62.8%) Type 1 + Type 2: 189 people (28.7%) Type 2: 56 people (8.5%);
Gonorrhea: 302 people (12.6%);
Syphilis: 80 people (3.3%);
HIV carriers: 58 people (2.4%);
Molluscum contagiosum: 58 people (2.4%);
Pubic lice: 21 people (0.9%);
Trichomonas vaginalis: 7 people (0.3%).
Among them, people with multiple sexually transmitted infections (n = 347 people, 14.5%); people with double sexually transmitted infections (n = 292 people, 84.1%); people with triple sexually transmitted infections (n = 47 people, 13.5%); people with four sexually transmitted infections (n = 7 people, 2%); People with severe STD infection (n = 1 person, 0.3%) = AIDS + syphilis + gonorrhea + herpes + chlamydia.
• In addition, AIDS combined with other STDs is (100%): AIDS (n = 58) AIDS + syphilis (n = 31, 53.4%) AIDS + cauliflower (n = 20, 34.5%) AIDS + gonorrhea (n = 13, 22.4%) AIDS + herpes (n = 7, 12.1%). Patients with multiple sexually transmitted infections are at least 2 to 5 times more likely to develop AIDS than the general population.
• This shows how important it is to quickly detect multiple sexually transmitted infections!
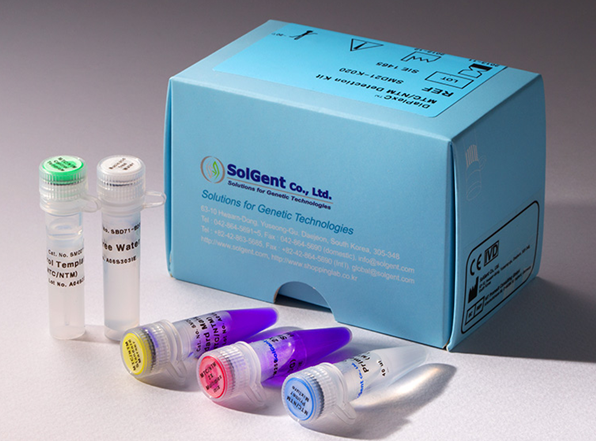
DiaPlexQ ™ STI-12 Detection Kit, CE; IVD is a broad-spectrum nucleic acid multiplex
NAT screening kit to detect common sexually transmitted infections
(Sexual-Transmitted Disease, STD).
This test suite is integrated
Human herpes virus type I & II (HSV-I & II),
C. Trachomatis,
N. Gonorrhoeae,
Trichomonas vaginalis (T. Vaginalis),
M. Genitalium,
Candida albicans (C. Albicans), etc. are seven common STD projects in Taiwan. Used with the Roche Cobas -480 Analyzer system, it can perform nucleic acid amplification reaction, cross-linking reaction and DNA signal generation at one time, effectively completing qualitative detection Testing and identification steps.
According to the original published reference, the positive predictive value (PPV) of this set was 96.6% -99.4%; the negative predictive value (NPV) was 98.2% -99.8%, compared with the Real Time-PCR related probe method value) is 0.93% -0.99%. The control group has no cross-reactivity with 41 other bacterial DNAs; the detection limit value (LOD) is 1 to 10 copies / reaction, which is a very good detection method. [Ref. Huh HJ, Ki C‐S, Yun SA, et al .. J Clin Lab Anal. 2019; 33: e22703.]. This set can also be combined with human papillomavirus genotyping (HPV Genotyping) for sampling and screening of the same brush tube
(HPV + STD 7 in One), in addition to shortening the experimental operation and reporting time and effectively reducing the risk of pollution, it can also reduce costs and price consumption, which is especially beneficial to women's health and health management. Early screening, early treatment, one more test, one more peace of mind!
• AIDS Quick Screening: Dual System Detection and Elimination (AIDS-HIV-Antigen / Antibody / Ag / Ab-Combo 15 minutes)
• Quick screening of syphilis: dual system detection and elimination method (Syphilis-Ag / Ab-RPR-TPPA 15 minutes)
• Neisseria. Gonorrhoeae
• Chlamydia. Trachomatis
• Candida. Albicans
• Trichomonas. Vaginalis
• Mycoplasma. Genitalium
• Human Herpes Virus Types I & II (Herpes-HSV-I & II)
• HPV Genotyping-human papillomavirus
• Collection process, blood sampling + urine collection + genital brushing sampling
• Test conditions, testing can only be performed after 4 weeks (28 days) of sexual contact
• Subject to conditions, AIDS, syphilis & abnormal items provide a review service
• Special conditions, urinary pruritus, pain and other symptoms should be prioritized for testing
• Report time, AIDS + syphilis completed in 15 minutes, and the remaining items will take about 4-5 working days
HPV + STD: 7 in One NAT PCR screening,
Multiple STD infections: 347 (14.5%);
Condyloma acuminatum: 772 people (32.3%);Chlamydia urethritis: 736 people (30.8%);
Genital herpes: 658 people (27.5%) Type 1: 413 people (62.8%) Type 1 + Type 2: 189 people (28.7%) Type 2: 56 people (8.5%);
Gonorrhea: 302 people (12.6%);
Syphilis: 80 people (3.3%);
HIV carriers: 58 people (2.4%);
Molluscum contagiosum: 58 people (2.4%);
Pubic lice: 21 people (0.9%);
Trichomonas vaginalis: 7 people (0.3%).
Among them, people with multiple sexually transmitted infections (n = 347 people, 14.5%); people with double sexually transmitted infections (n = 292 people, 84.1%); people with triple sexually transmitted infections (n = 47 people, 13.5%); people with four sexually transmitted infections (n = 7 people, 2%); People with severe STD infection (n = 1 person, 0.3%) = AIDS + syphilis + gonorrhea + herpes + chlamydia.
• In addition, AIDS combined with other STDs is (100%): AIDS (n = 58) AIDS + syphilis (n = 31, 53.4%) AIDS + cauliflower (n = 20, 34.5%) AIDS + gonorrhea (n = 13, 22.4%) AIDS + herpes (n = 7, 12.1%). Patients with multiple sexually transmitted infections are at least 2 to 5 times more likely to develop AIDS than the general population.
• This shows how important it is to quickly detect multiple sexually transmitted infections!
DiaPlexQ ™ STI-12 Detection Kit, CE; IVD is a broad-spectrum nucleic acid multiplex
NAT screening kit to detect common sexually transmitted infections
(Sexual-Transmitted Disease, STD).
This test suite is integrated
Human herpes virus type I & II (HSV-I & II),
C. Trachomatis,
N. Gonorrhoeae,
Trichomonas vaginalis (T. Vaginalis),
M. Genitalium,
Candida albicans (C. Albicans), etc. are seven common STD projects in Taiwan. Used with the Roche Cobas -480 Analyzer system, it can perform nucleic acid amplification reaction, cross-linking reaction and DNA signal generation at one time, effectively completing qualitative detection Testing and identification steps.
According to the original published reference, the positive predictive value (PPV) of this set was 96.6% -99.4%; the negative predictive value (NPV) was 98.2% -99.8%, compared with the Real Time-PCR related probe method value) is 0.93% -0.99%. The control group has no cross-reactivity with 41 other bacterial DNAs; the detection limit value (LOD) is 1 to 10 copies / reaction, which is a very good detection method. [Ref. Huh HJ, Ki C‐S, Yun SA, et al .. J Clin Lab Anal. 2019; 33: e22703.]. This set can also be combined with human papillomavirus genotyping (HPV Genotyping) for sampling and screening of the same brush tube
(HPV + STD 7 in One), in addition to shortening the experimental operation and reporting time and effectively reducing the risk of pollution, it can also reduce costs and price consumption, which is especially beneficial to women's health and health management. Early screening, early treatment, one more test, one more peace of mind!
• AIDS Quick Screening: Dual System Detection and Elimination (AIDS-HIV-Antigen / Antibody / Ag / Ab-Combo 15 minutes)
• Quick screening of syphilis: dual system detection and elimination method (Syphilis-Ag / Ab-RPR-TPPA 15 minutes)
• Neisseria. Gonorrhoeae
• Chlamydia. Trachomatis
• Candida. Albicans
• Trichomonas. Vaginalis
• Mycoplasma. Genitalium
• Human Herpes Virus Types I & II (Herpes-HSV-I & II)
• HPV Genotyping-human papillomavirus
• Collection process, blood sampling + urine collection + genital brushing sampling
• Test conditions, testing can only be performed after 4 weeks (28 days) of sexual contact
• Subject to conditions, AIDS, syphilis & abnormal items provide a review service
• Special conditions, urinary pruritus, pain and other symptoms should be prioritized for testing
• Report time, AIDS + syphilis completed in 15 minutes, and the remaining items will take about 4-5 working days
HPV + STD: 7 in One NAT PCR screening,
Eliminate all pathogens of multiple sexually transmitted diseases



